 
This is due to electrons having a really small mass, therefore not significantly contributing to the atomic mass. You can calculate the atomic mass by adding the number of neutrons and protons in the atom. Average masses are generally expressed in unified atomic mass units (u), where 1 u is equal to exactly one-twelfth the mass of a neutral atom of carbon-12. Because the sum of the numbers of protons and neutrons equals the mass number, 127, the number of neutrons is 74 (127 − 53 = 74). The easiest way to find the atomic mass of an element, is to look on the periodic table. The average atomic mass (sometimes called atomic weight) of an element is the weighted average mass of the atoms in a naturally occurring sample of the element. The average Atomic Mass of this isotope is 17.999 or 18 (approx.). And thus the sum of these two will result into 18, which will be the Atomic Mass of that certain atom of oxygen Isotopes in amu terms (Atomic Mass units). 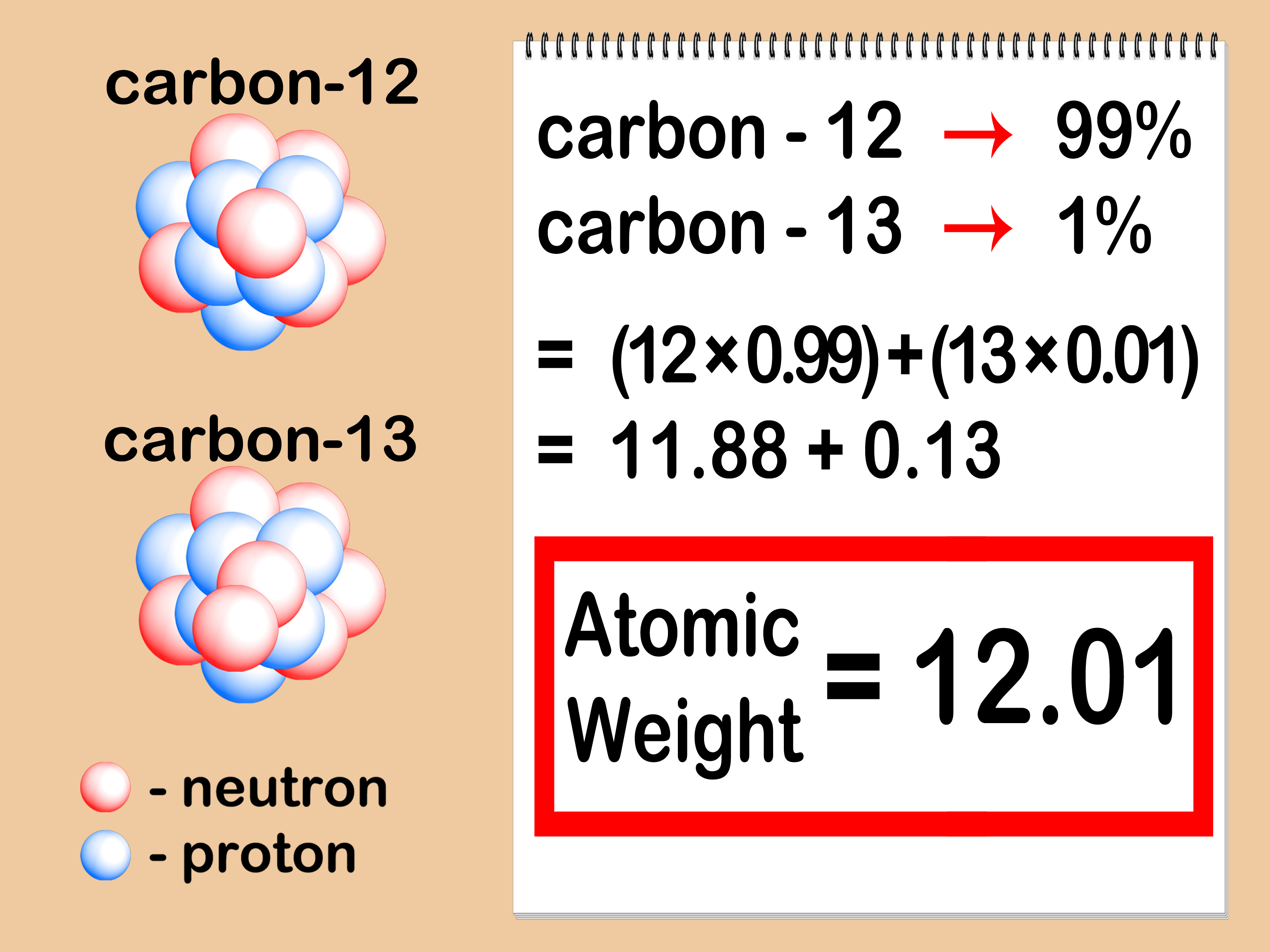 
The atomic number of iodine (53) tells us that a neutral iodine atom contains 53 protons in its nucleus and 53 electrons outside its nucleus. The student will be able to find via the periodic table that the atoms of oxygen have 8 protons and 8 electrons in them. Determine the numbers of protons, neutrons, and electrons in one of these iodine anions. The iodine atoms are added as anions, and each has a 1− charge and a mass number of 127. Fortunately, you do not need to memorize this value, because you can easily calculate its value from Avogadros number, NA, which you are expected to know: 1u 1 NA g 1 1000 NA Kg (4.1.3) (4.1.3) 1 u 1 N A g 1 1000 N A K g. The addition of small amounts of iodine to table salt (iodized salt) has essentially eliminated this health concern in the United States, but as much as 40% of the world’s population is still at risk of iodine deficiency. The unified atomic mass unit is defined as 1/12 of the mass of one atom of carbon-12. (credit a: modification of work by “Almazi”/Wikimedia Commons credit b: modification of work by Mike Mozart) (b) The addition of small amounts of iodine to salt, which prevents the formation of goiters, has helped eliminate this concern in the US where salt consumption is high. \): (a) Insufficient iodine in the diet can cause an enlargement of the thyroid gland called a goiter. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
